Towards a Hepatitis C Vaccine - Mouse Model Developed
Worldwide, an estimated 400,000 people die annually from chronic infection with hepatitis C virus (HCV). A vaccine is still not available. One obstacle to developing vaccines is the lack of animal models as HCV only infects humans . As part of an international research collaboration, Scientists from the Paul-Ehrlich-Institut have identified barriers in murine liver cells that prevent HCV infection of rodents. This is an important step in the development of a permissive mouse model for preclinical testing of HCV vaccine candidates and might well accelerate their development. Science Advances reports on the investigations in its online edition of 04 November 2020.
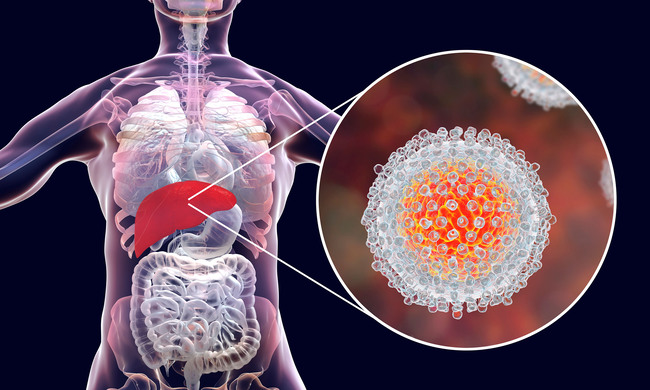
About 71 million people worldwide are chronically infected with the hepatitis C virus (HCV). Chronic liver inflammation can cause progressive liver damage and lead to serious complications including liver cirrhosis and liver cancer. HCV naturally infects only humans. This is because viruses are usually well adapted to their host and cross-species transmission is limited by host factor incompatibilities and restriction factors. On the one hand, the restricted host range presents an opportunity for complete eradication of the virus through effective vaccination, as no animal reservoir exists. At the same time, however, it has the disadvantage that no animal models are available with which to study the immunological processes underlying chronic infection. Furthermore, there is no possibility to test new therapeutic medicines for the treatment of HCV infections or vaccine candidates in animal models.
So far, the barriers limiting HCV infection to humans have only been partially identified. Scientists led by Dr Richard Brown, Research Group Leader in the Division of Veterinary Medicine at the Paul-Ehrlich-Institut, and Professor Thomas Pietschmann, Head of the Department of Experimental Virology at Twincore, Centre for Experimental and Clinical Infection Research, Hanover, have joined forces with research teams from Germany, the Netherlands, Sweden, Great Britain, USA, Spain and Belgium to search specifically for the barriers that have so far made it impossible for the hepatitis C virus to infect the liver of mice. Also involved in the project was a team led by Professor Charles Rice, who was awarded the Nobel Prize in Physiology or Medicine in 2020 for the discovery of the HCV virus. The mouse was the focus of the research team because it is very well suited as an animal model for research into vaccines against HCV – provided the virus can infect the liver.
Using state-of-the-art methods, the research team identified proteins in the liver cells of mice that contribute to preventing HCV infection. They reported an unexpected interaction between a lectin and a complement receptor, which cooperate to mediate protection against HCV infection of mice. Lectins are a group of specific proteins that bind to a variety of carbohydrate structures and can thus influence various biological responses. The complement system is a component of the immune system which specifically targets pathogens. Switching off the genes that express these proteins in the liver made the mice more susceptible to HCV infection. These results pave the way for the development of mouse models that could be used to study the disease progression associated with chronic hepatitis C infection and develop effective and safe vaccines.
Original Publication
Brown RJP, Tegtmeyer B, Sheldon J, Khera T, Anggakusuma, Todt D, Vieyres G, Weller R, Joecks S, Zhang Y, Sake S, Bankwitz D, Welsch K, Ginkel C, Engelmann M, Gerold G, Steinmann E, Yuan Q, Ott M, Vondran FWR, Krey T, Ströh LJ, Miskey C, Ivics Z, Herder V, Baumgärtner W, Lauber C, Seifert M, Tarr AW, McClure CP, Randall G, Baktash Y, Ploss A, Thi VLD, Michailidis E, Saeed M, Verhoye L, Meuleman P, Goedecke N, Wirth D, Rice CM, Pietschmann T (2020): Liver-expressed Cd302 and Cr1l limit hepatitis C virus cross-species transmission to mice.
Sci Adv 6: eabd3233.
Online-Abstract
top



