Compassionate Use Programmes
Legal Basis
"Compassionate Use" was introduced into the German legislation with the 14th amendment of the German Medicines Act (AMG) and modified by the amendment of the German Medicines Act in July 2009.
The procedure for authorisation and placing on the market of new medicinal products on national level is regulated in Section 21 of the Law on Medicinal Products and on European level on the basis of Directive 2001/83/EC in conjunction with Article 83 of Regulation (EC) No. 726/2004. According to these regulations the use of unauthorized medicinal products is only possible in clinical trials but not for placing on the market. As an exception, according to Article 83 of EC No. 726/2004 unauthorised medicinal products may be placed at the disposal of severely diseased people as long as this is in line with a compassionate use and there is no therapeutic alternative.
The Ordinance on Medicinal Products for Compassionate Use (AMHV) pursuant to Section 80 of the German Medicines Act was published on 21 July 2010 in the Federal Law Gazette 2010 part I No 37 and came into force on 22 July 2010. A non-official translation is available for download.
Ordinance on Medicinal Products for Compassionate Use – AMHV
Requirements for compassionate use
The Ordinance on Medicinal Products for Compassionate Use (AMHV) came into force on 22nd July 2010. The AMHV regulates the procedure for the placing on the market of unauthorized medicinal product in order to treat at no charge a specific group of severe diseased patients, who cannot be treated with an authorized medicinal product and an application for marketing authorisation has been submitted or a clinical trial is being conducted on it. The administration of an unauthorised medicinal product to an individual patient under the direct supervision of a doctor is not in the scope of the AMHV.
The AMHV introduces the confirmed notification procedure for compassionate use programmes by the competent authorities. According to Section 77 of the German Medicinal Products Act, the Bundesinstitut für Arzneimittel und Medizinprodukte and the Paul-Ehrlich Institut are also the competent authorities for compassionate use programmes.
Notification of a Compassionate Use Programme
For decision of applicability of the compassionate use programme please follow the flow chart:
Flow chart for Compassionate Use Programmes
For notification please use the forms below.
The notification and the required documents shall be made in hard copy and electronically.
Please submit compassionate use programmes in the responsibility of the Paul-Ehrlich-Institut to the following address:
Paul-Ehrlich-Institut
Referat Klinische Prüfungen
Paul-Ehrlich-Str. 51-59
63225 Langen
Tel +49 6103 77 1810
For more information please contact: ct@pei.de
Please note: Since 23 July 2010 the commencement of any new compassionate use programme requires a confirmation of notification by the higher federal competent authority (in this case: Paul-Ehrlich-Insitut).
Timelines for Notifications of Compassionate Use Programmes for different categories of medicinal products in the responsibility of the Paul-Ehrlich-Institut
Category 1 – Timeline 1:
Products known from clinical trials or applications for marketing authorisation (incl. Genetically Modified Organisms, without changes capable of modifying the assessment of the risk to the health of third parties and the environment; (except Advanced Therapy Medicinal Products)
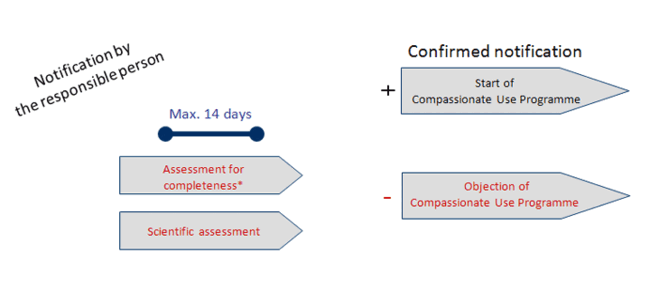
 * Documents formally insufficient please see timeline 4 (click to enlarge).
Source: Paul-Ehrlich-Institut
* Documents formally insufficient please see timeline 4 (click to enlarge).
Source: Paul-Ehrlich-Institut
Category 2 – Timeline 2:
- Products unknown from clinical trials or applications for marketing authorisation
or - The Medicinal product is an Advanced Therapy Medicinal Product (known or unknown)
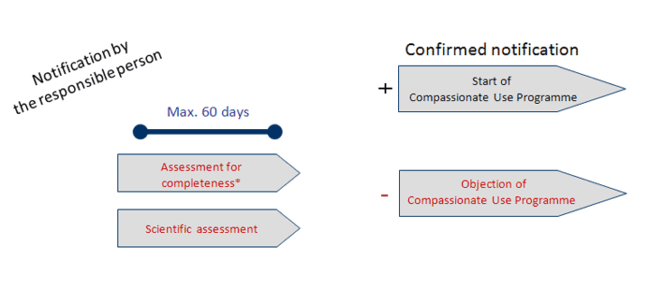
 * Documents formally insufficient please see timeline 4 (click to enlarge).
Source: Paul-Ehrlich-Institut
* Documents formally insufficient please see timeline 4 (click to enlarge).
Source: Paul-Ehrlich-Institut
Category 3 – Timeline 3:
- Genetically Modified Organisms, unknown from clinical trials or application for marketing authorisation
or - Known Genetically Modified Organisms, with changes capable of modifying the assessment of the risk to the health of third parties and the environment
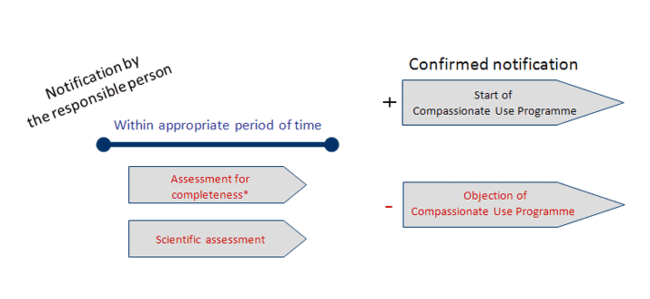
 * Documents formally insufficient please see timeline 4 (click to enlarge).
Source: Paul-Ehrlich-Institut
* Documents formally insufficient please see timeline 4 (click to enlarge).
Source: Paul-Ehrlich-Institut
Categories 1,2,3 – Timeline 4:
Formally insufficient documents

 *There is no timeline for additional documents (click to enlarge).
Source: Paul-Ehrlich-Institut
*There is no timeline for additional documents (click to enlarge).
Source: Paul-Ehrlich-Institut
top






