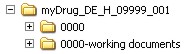Submission in eCTD Format
Submission in eCTD Format
The Paul Ehrlich Institute (PEI) prefers electronic submissions for all procedures in the eCTD format, because only in this format can the lifecycle of the dossier be mapped accordingly.
If a product is submitted in the eCTD format, all subsequent submissions are expected in this format. A mixed submission eCTD / NeeS is not possible, this would render a lifecycle management of the documentation impossible ("once eCTD always eCTD").
If a change ofsubmission format is made from paper or NeeS to eCTD (e.g. on the occasion of a marketing authorisation, a renewal or a change notification (variation)), you are requested to submit a Baseline (Sequence 0000) for this product. It comprises at least modules 1 and 3 and is a great help for the evaluation process.
Structure and naming of the eCTD Directory
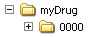
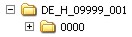
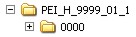
The authorisation number should be indicated in the name of the root directory, the name of the drug or the procedure number or both or, as a further alternative.. The name should provide informational value that takes into account the product structure.
Preferred Name
 Source: PEI
Source: PEI
Alternative Options
 Source: PEI
Source: PEI
 Source: PEI
Source: PEI
 Source: PEI
Source: PEI
Below the root directory, the sequence folders of the submission (<sequence>) as well as separate folders for so-called working documents (<sequence> -working documents) are created. These correspond to the minimum electronic submission documents specified in the AMG-EV (in accordance with module 1.3.1 and module 2) and must be submitted as rtf documents.
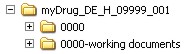 Source: PEI
Source: PEI
If the submission concerns several applications for authorisations or granted authorisations (e.g. for a worksharing procedure), the appropriate directories must be set up in accordance with the above-mentioned naming scheme.
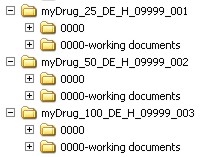 Source: PEI
Source: PEI
If several sequences are submitted in one submission step, the appropriate directories for each sequence have to be created using the following pattern:
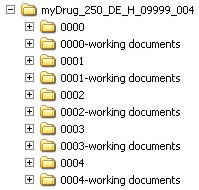 Source: PEI
Source: PEI
Subfolders for the modules, a util directory with the schemes used, a valid XML backbone and the associated md5 checksum must be present within the sequence.
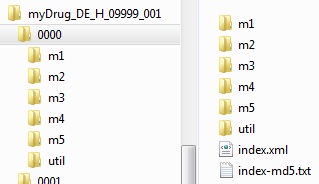 Source: PEI
Source: PEI
EU Module 1 - Content of EU Envelope
For all eCTD submissions, the following elements and attributes must be filled in the XML structure for module 1 within the eu envelope:
| Element | Attribut | Beispiel |
| submission | type | var-type2 |
| number | | EMA/H/xxxx/WS/001 |
| tracking number | | EMA/H/C/0123/WS005/045 |
| applicant | | PharmaCompany Ltd. |
| agency | code | DE-PEI |
| invented-name | | myDrug |
| sequence | | 0000 |
| submission-description | | Responses to Day 120 List of Questions |
top
Electronic Submission in NeeS Format
Electronic Submission in NeeS Format
The Paul-Ehrlich-Institut (PEI) prefers submissions in the eCTD format, because only in this format can the lifecycle of the dossier can be mapped accordingly. In addition, however, electronic submissions will continue to be accepted using the NeeS Guideline, i.e .without the XML backbone of the eCTD format.
Please note that the NeeS format cannot be used in the centralised procedure.
When changing the submission format from paper to NeeS (e.g. on the occasion of amarketing authorisation, a renewal or a change notification (variation)), you are requested to submit a so-called baseline (Sequence 0000) for this product. It comprises at least modules 1 and 3 and is a great help for the evaluation process.
Structure and naming of the NeeS directory structure
The authorisation number should be indicated in the name of the root directory, the name of the drug or the procedure number or both or, as a further alternative to the authorisation number. The name should provide informational value that takes into account the product structure.
Preferred Name
 Source: PEI
Source: PEI
Alternative Options
 Source: PEI
Source: PEI
 Source: PEI
Source: PEI
 Source: PEI
Source: PEI
Below the root directory, the sequence folders of the submission (<sequence>) are created as well as separate folders for so-called working documents (<sequence> working documents). These correspond to the minimum electronic submission documents specified in the AMG-EV (in accordance with module 1.3.1 and module 2) and must be submitted as rtf documents.
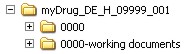 Source: PEI
Source: PEI
If the submission concerns several applications for authorisations or granted authorisations (e.g. for a worksharing procedure), the appropriate directories must be set up in accordance with the above-mentioned naming scheme.
 Source: PEI
Source: PEI
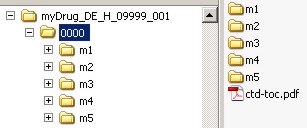
If several sequences are submitted in one submission step, the appropriate directories for each sequence have to be created using the following pattern:
 Source: PEI
Source: PEI
In this context, the sequential numbering of the sequences of a dossier is also recommended. This avoids problems with the timing in the evaluation process.
Within the sequence, subfolders for the modules must be present, and also a table of contents "ctd-toc.pdf".
 Source: PEI
Source: PEI
top
Electronic submission in vNeeS format
Electronic submission in vNeeS format
The Paul-Ehrlich-Institut (PEI) welcomes the use of the vNeeS format for submissions in the field of immunological veterinary medicinal products.
Information on the structuring and formatting of submissions in the vNeeS format is available on the eSubmission website of the European Medicines Agency (EMA).
When changing the submission format from paper to vNeeS (e.g. on the occasion of amarketing authorisation, a renewal or change notification (variation) you are requested to submit a so-called Baseline (Sequence 0000) for this product. It comprises at least parts 1 and 2 and is a great help for the evaluation process.
Structure and naming of the vNeeS directory structure
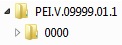
The authorisation number should be indicated in the name of the root directory, the name of the drug or the procedure number or both or, as a further alternative.. The name should informatively consider the product structure.
Preferred Name
 Source: PEI
Source: PEI
Alternative Options
 Source: PEI
Source: PEI
 Source: PEI
Source: PEI
 Source: PEI
Source: PEI
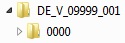
Below the root directory, the sequence folders of the submission (<sequence>) are created.
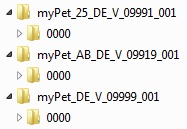
If the submission concerns several applications for authorisation or granted authorisations (e.g. for a worksharing procedure), corresponding directories must be set up in accordance with the above-mentioned designation scheme.
 Source: PEI
Source: PEI
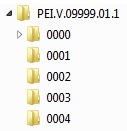
If several sequences are submitted at once, the appropriate directories for each sequence must be created using the following pattern:
 Source: PEI
Source: PEI
In this context, the sequential numbering of the sequences of a dossier is also recommended. This avoids problems with the timing of submissions as part of the evaluation process.
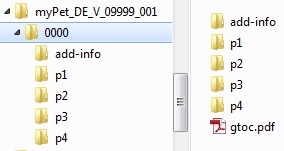
Within the sequence, subfolders for the modules must be present for additional working documents, as well as a table of contents "gtoc.pdf".
 Source: PEI
Source: PEI
top
Technical Validation
Technical Validation
Before submitting a dossier, we recommend that you carry out a technical validation. This will allow you to determine whether your dossier actually meets the requirements mentioned above (e.g. ICH M2 eCTD v.3.2.2, EU M1 v.2.0, file and folder structure and naming conventions, PDF configurations), or whether you have to fix any errors before.
Several software vendors are offering free validation programs that meet the current specifications for eCTD (EU M1 v2.0 and ICH eCTD v. 3.2.2, EU eCTD Validation Criteria v5.0), NeeS (EU NeeS Validation Criteria v4.0 ) and vNeeS (vNeeS Validation Checklist v2.2). Unfortunately, technical support for the use of these validators cannot be provided by the Paul Ehrlich Institut (PEI).
In any case, the PEI will carry out a technical validation by itself. Applicants are only informed of the result if errors have occurred as part of the pass / fail criteria. Entering your email address on submission will facilitate the submission of the validation report. Best practice warnings are not communicated.
Errors will cause failure to import a submission, and you, the applicant, will be asked to correct the errors and resubmit the sequence.
Warnings in the best practice area are tolerated. However, they indicate that predetermined requirements are not met.
Deadlines can only be started after a successful technical validation.
top